what type of reaction is necessary to produce a dipeptide from individual amino acids?

In organic chemical science, a peptide bail is an amide type of covalent chemic bond linking two consecutive blastoff-amino acids from C1 (carbon number one) of one alpha-amino acid and N2 (nitrogen number two) of another, along a peptide or protein chain.[1]
It tin also be called a eupeptide bond [one] to distinguish it from an isopeptide bail, which is another blazon of amide bail betwixt two amino acids.
Synthesis [edit]

When two amino acids class a dipeptide through a peptide bond,[ane] it is a type of condensation reaction.[2] In this kind of condensation, 2 amino acids approach each other, with the non-side chain (C1) carboxylic acid moiety of 1 coming near the not-side chain (N2) amino moiety of the other. 1 loses a hydrogen and oxygen from its carboxyl group (COOH) and the other loses a hydrogen from its amino grouping (NH2). This reaction produces a molecule of water (H2O) and two amino acids joined by a peptide bond (-CO-NH-). The two joined amino acids are called a dipeptide.
The amide bail is synthesized when the carboxyl group of one amino acid molecule reacts with the amino group of the other amino acid molecule, causing the release of a molecule of h2o (HiiO), hence the process is a dehydration synthesis reaction.
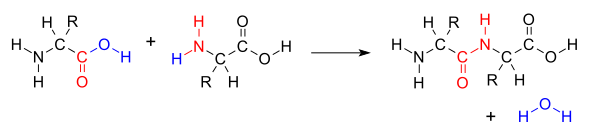
The aridity condensation of two amino acids to form a peptide bond (carmine) with expulsion of water (blueish).
The formation of the peptide bail consumes energy, which, in organisms, is derived from ATP.[3] Peptides and proteins are bondage of amino acids held together by peptide bonds (and sometimes by a few isopeptide bonds). Organisms use enzymes to produce nonribosomal peptides,[4] and ribosomes to produce proteins via reactions that differ in details from aridity synthesis.[5]
Some peptides, like blastoff-amanitin, are called ribosomal peptides equally they are made past ribosomes,[half dozen] but many are nonribosomal peptides as they are synthesized by specialized enzymes rather than ribosomes. For instance, the tripeptide glutathione is synthesized in 2 steps from free amino acids, by two enzymes: glutamate–cysteine ligase (forms an isopeptide bail, which is not a peptide bond) and glutathione synthetase (forms a peptide bond).[7] [8]
Degradation [edit]
A peptide bail can exist broken by hydrolysis (the addition of water). The hydrolysis of peptide bonds in water releases 8–16 kilojoule/mol (2–4 kcal/mol) of Gibbs free energy.[9] This process is extremely tiresome, with the half life at 25 °C of betwixt 350 and 600 years per bond.[x]
In living organisms, the process is normally catalyzed by enzymes known as peptidases or proteases, although in that location are reports of peptide bond hydrolysis acquired by conformational strain as the peptide/protein folds into the native structure.[11] This non-enzymatic procedure is thus not accelerated by transition state stabilization, simply rather by ground state destabilization.
Spectra [edit]
The wavelength of absorption for a peptide bond is 190–230 nm[12] (which makes it particularly susceptible to UV radiation).
Cis/trans isomers of the peptide group [edit]
Significant delocalisation of the solitary pair of electrons on the nitrogen atom gives the group a partial double bond character. The partial double bond renders the amide group planar, occurring in either the cis or trans isomers. In the unfolded state of proteins, the peptide groups are gratuitous to isomerize and adopt both isomers; however, in the folded country, simply a unmarried isomer is adopted at each position (with rare exceptions). The trans class is preferred overwhelmingly in most peptide bonds (roughly 1000:1 ratio in trans:cis populations). Even so, X-Pro peptide groups tend to have a roughly 30:1 ratio, presumably considering the symmetry between the and atoms of proline makes the cis and trans isomers well-nigh equal in energy (see figure, beneath).
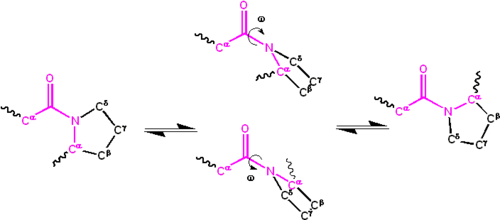
Isomerization of an X-Pro peptide bail. Cis and trans isomers are at far left and far right, respectively, separated past the transition states.
The dihedral angle associated with the peptide group (defined past the four atoms ) is denoted ; for the cis isomer (synperiplanar conformation) and for the trans isomer (antiperiplanar conformation). Amide groups tin isomerize about the C'-N bond betwixt the cis and trans forms, albeit slowly ( 20 seconds at room temperature). The transition states requires that the fractional double bond exist cleaved, so that the activation free energy is roughly fourscore kilojoule/mol (20 kcal/mol). Still, the activation energy can be lowered (and the isomerization catalyzed) past changes that favor the single-bonded form, such as placing the peptide group in a hydrophobic environment or donating a hydrogen bond to the nitrogen cantlet of an Ten-Pro peptide group. Both of these mechanisms for lowering the activation energy have been observed in peptidyl prolyl isomerases (PPIases), which are naturally occurring enzymes that catalyze the cis-trans isomerization of X-Pro peptide bonds.
Conformational protein folding is usually much faster (typically ten–100 ms) than cis-trans isomerization (10–100 due south). A nonnative isomer of some peptide groups tin can disrupt the conformational folding significantly, either slowing it or preventing information technology from even occurring until the native isomer is reached. However, not all peptide groups take the same effect on folding; nonnative isomers of other peptide groups may not bear upon folding at all.
Chemic reactions [edit]
Due to its resonance stabilization, the peptide bond is relatively unreactive nether physiological conditions, fifty-fifty less than similar compounds such every bit esters. Nevertheless, peptide bonds tin undergo chemic reactions, commonly through an assault of an electronegative cantlet on the carbonyl carbon, breaking the carbonyl double bond and forming a tetrahedral intermediate. This is the pathway followed in proteolysis and, more than generally, in N-O acyl exchange reactions such as those of inteins. When the functional group attacking the peptide bond is a thiol, hydroxyl or amine, the resulting molecule may be called a cyclol or, more specifically, a thiacyclol, an oxacyclol or an azacyclol, respectively.
See besides [edit]
- The Proteolysis Map
References [edit]
- ^ a b c "Nomenclature and Symbolism for Amino Acids and Peptides. Recommendations 1983". European Periodical of Biochemistry. 138 (1): 9–37. 1984. doi:10.1111/j.1432-1033.1984.tb07877.10. ISSN 0014-2956. PMID 6692818.
- ^ Muller, P (1994-01-01). "Glossary of terms used in concrete organic chemistry (IUPAC Recommendations 1994)". Pure and Applied Chemistry. 66 (five): 1077–1184. doi:10.1351/pac199466051077. ISSN 1365-3075.
- ^ Watson J, Hopkins N, Roberts J, Agetsinger Steitz J, Weiner A (1987) [1965]. Molecualar Biology of the Gene (hardcover) (4th ed.). Menlo Park, CA: The Benjamin/Cummings Publishing Company, Inc. p. 168. ISBN978-0805396140.
- ^ Miller BR, Gulick AM (2016). "Structural Biology of Nonribosomal Peptide Synthetases". Methods in Molecular Biology. 1401: three–29. doi:x.1007/978-i-4939-3375-4_1. ISBN978-1-4939-3373-0. PMC4760355. PMID 26831698.
- ^ Griffiths AJ, Miller JH, Suzuki DT, Lewontin RC, Gelbart WM (2000). Protein synthesis. An Introduction to Genetic Analysis (7th ed.). New York: Westward. H. Freeman. ISBN978-0716735205.
- ^ Walton JD, Hallen-Adams HE, Luo H (2010). "Ribosomal biosynthesis of the circadian peptide toxins of Amanita mushrooms". Biopolymers. 94 (5): 659–64. doi:ten.1002/bip.21416. PMC4001729. PMID 20564017.
- ^ Wu 1000, Fang YZ, Yang South, Lupton JR, Turner ND (March 2004). "Glutathione metabolism and its implications for health". The Periodical of Nutrition. 134 (3): 489–92. doi:10.1093/jn/134.3.489. PMID 14988435.
- ^ Meister A (November 1988). "Glutathione metabolism and its selective modification". The Journal of Biological Chemistry. 263 (33): 17205–eight. PMID 3053703.
- ^ Martin RB (December 1998). "Free energies and equilibria of peptide bond hydrolysis and formation". Biopolymers. 45 (5): 351–353. doi:10.1002/(SICI)1097-0282(19980415)45:v<351::Assistance-BIP3>3.0.CO;2-Grand.
- ^ Radzicka A, Wolfenden R (1996-01-01). "Rates of Uncatalyzed Peptide Bond Hydrolysis in Neutral Solution and the Transition State Affinities of Proteases". Journal of the American Chemical Order. 118 (26): 6105–6109. doi:x.1021/ja954077c. ISSN 0002-7863.
- ^ Sandberg A, Johansson DG, Macao B, Härd T (April 2008). "Sea domain autoproteolysis accelerated by conformational strain: energetic aspects". Periodical of Molecular Biological science. 377 (four): 1117–29. doi:10.1016/j.jmb.2008.01.051. PMID 18308334.
- ^ Goldfarb AR, Saidel LJ, Mosovich E (Nov 1951). "The ultraviolet absorption spectra of proteins". The Journal of Biological Chemistry. 193 (1): 397–404. PMID 14907727.
Source: https://en.wikipedia.org/wiki/Peptide_bond








0 Response to "what type of reaction is necessary to produce a dipeptide from individual amino acids?"
Post a Comment